Studies have shown that monovalent copper ions (Cu+) eradicate bacteria and fungi even at low concentrations (0.2 ppm). Monovalent copper ions were found to be much more active than monovalent silver ions.
Studies have shown that monovalent copper ions (Cu+) eradicate bacteria and fungi even at low concentrations (0.2 ppm). Monovalent copper ions were found to be much more active than monovalent silver ions. Divalent copper ions and metallic copper were not found to be active at all [1, 2, 3].
The reduction of divalent copper ions to monovalent copper ions can be achieved in an aqueous solution using copper-containing electrodes with a 0.3V potential difference between them. An effective concentration (for eradicating bacteria and fungi) of monovalent copper ions in water can be maintained over time using the above electrochemical system. We successfully utilized the system to disinfect water in large volumes in an open-air atmosphere. This study presents a method for water disinfection, free from chlorine or bromine, using only 0.004mM (~0.2ppm) Cu ions at natural pH with no other additives. The article presents results from a 20 m3 research pool, showing a 5 log unit decrease in the bacterial population 10 min from adding the bacteria, and 30 min to reach zero bacteria.
Copper is an essential intracellular element in trace concentrations due to the ability of copper to exist in ionic forms, alternating between cuprous (Cu+), cupric (Cu2+), and (Cu3+) oxidation states. In high concentration, however, it is toxic [4].
Copper’s been exploited since ancient times for medicinal purposes [5]. Currently, copper is used as a water purifier, fungicide, and bactericide. Introducing copper into cotton fibers, polymeric materials, and clothing as a means of bolstering their biocidal properties was suggested more than a decade ago [6,7,8]. Copper surfaces help limit the spread of multidrug-resistant bacteria in hospitals [9]. In all of the above uses, the lack of regularity in the results is reported; the decrease in the microorganism population is not always obtained. All this makes understanding the processes resulting in the potent antibacterial effect of copper highly relevant.
Our studies have shown that monovalent copper ions (Cu+) have dramatic antibacterial and antifungal activity compared to Cu+2 ions or metallic copper surfaces, which do not show such properties [1,2]. The proposed mechanism involves the entry of monovalent copper ions through the bacterial cell membrane (via sodium channels) and the paralysis of enzymatic systems in the cytoplasm [10]. Although the monovalent copper ion reacts rapidly with molecular oxygen [11] and undergoes disproportionation in water [11], we have reported formulations and solutions that maintain an effective antibacterial concentration of monovalent copper ions in the room atmosphere [12, 13].
As aforesaid in aqueous solutions, the common oxidation state of copper ions is bivalent (Cu2+, cupric). Copper in the monovalent state (Cu+, cuprous) remains in a low to negligible concentration due to a disproportion reaction (self-oxidation of monovalent copper to divalent copper and metallic copper), and due to rapid oxidation by molecular oxygen to divalent copper. In systems where the environment supported the reduction of divalent to monovalent copper ions, the antimicrobial effect was enhanced [1,2,12].
To obtain and maintain an effective concentration of monovalent copper ions in a divalent copper ions solution, the divalent copper ions must first be reduced to monovalent copper ions, and second, to prevent disproportionation in water, substances (ligands) that stabilize the monovalent copper ions over the divalent ones can be added to the water. Acetonitrile [14], benzoic acid [15], and ATP [16, 17] are good examples of Cu+ stabilizing reagents that shift the existing equilibrium between oxidation states to the formation of two Cu+ ions from one Cu2+ ion and metallic copper.
Our previous research [1,2] was successful in exploiting this technique of Cu+ ion production, which then led to a series of studies devoted to investigating the antimicrobial effect of monovalent copper. We have clearly shown the superior efficacy of Cu+ ions over Cu2+ ions in killing E. coli and Staphylococcus aureus bacteria, and 15 more species of bacteria and fungi. Moreover, our studies have shown that Cu+ ions have a substantially higher efficacy than Ag+ ions, which are currently widely used as an antibacterial agent [1]. The antibacterial activity of Cu+ ions was found to be intensified by a high temperature, a low molecular oxygen concentration, a low pH, and a poor carbon source [2]. On the whole, our findings suggest that Cu+ should be considered as a potent antimicrobial agent.
The present article reports the creation of an effective concentration of monovalent copper ions in aqueous solutions exposed to air and utilizing this ability to disinfect water.
Cup experiments (up to 250 ml): The system consists of a beaker with a volume of 100 to 250 ml, a pair of electrodes with a gap of 0.5 cm between them and a water-immersed area of 2 cm3. A DC low-voltage laboratory power supply provides current to electrodes at a constant potential. Aqueous solution contained different concentrations of copper ions originating from copper (CuSO4, CuCl2) salts dissolved in tab water (~500ms).
20 cubic meters research pool: The system consists 20 m3 plastic pool with circulation system with flow rate of 4 cubic meters per hour, send filter and control panel (pH, ORP, conductivity, temperature, turbidity) with data logger and internet connection.
Wastewater disinfection: The system consists sand filter, receiving tank, feed pump, copper ion-charged cation exchanger, electrochemical cell, circulated intermediate tank, circulation pump with copper ion-charged cation exchanger and electrochemical cell, product tank.
Ion exchanger: Aminophosphonic acid chelating resin, such as Purolite S940 or Aldex 212, charged with 30-60 mg Cu2+ per gram resin.
Determination of copper ion concentration: To determine the total copper ion concentration, ICP- Inductively coupled plasma atomic emission spectroscopy (SPECTRO ARCOS) and light absorption, using Spectrophotometer, at a wavelength of 600 nm of a copper ion Cuprizone complex were used against a calibration curve.
Counting colony-forming units (CFU): For estimates of the number of bacteria or fungal cells in a sample, we used colony-forming unit (CFU) counts. Bacteria were counted using a routine CFU technique, i.e., by plating bacteria from serial dilutions onto LB agar and incubating overnight at 37 oC for bacteria an YPD agar at 30 oC for fungus.
Cup experiments (up to 250 ml)
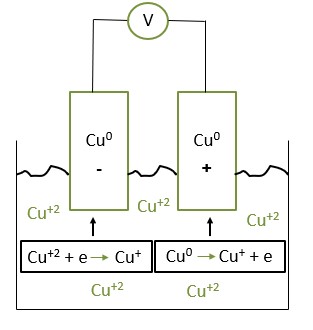
Figure 1 shows a schematic description of the experimental system. E. Coli bacteria population, at logarithmic phase, was introduced into the aqueous solution at various concentrations. During the experiment, the bacterial population was monitored over time using the CFU method, along with the concentration of copper ions and pH.
Figure 1: Schematic description of a laboratory experimental system
When both electrodes are made of copper, and the aqueous solution has an initial concentration of 0.2 to 10ppm Cu2+ and 104/ml E. Coli bacteria, a potential of 0.3V causes the eradication of the bacterial population (Figure 2).
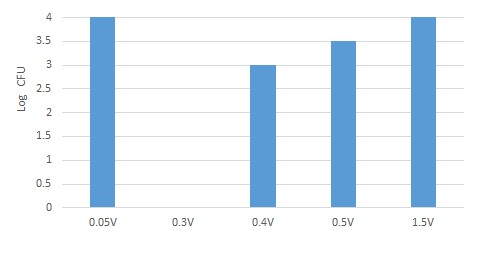
Figure 2: Bar chart, X-axis, potential in Volt between the electrodes, Y-axis, E. Coli bacteria in log CFU/ml units approximately three hours from the start of the experiment. Initial concentration 10ppm Cu+2, pH ~7.
The results shown in Figure 2 show that only a certain voltage (corresponding to the reduction of divalent copper to monovalent) causes damage to the bacterial population. A higher voltage will reduce the divalent copper ions to metal copper.
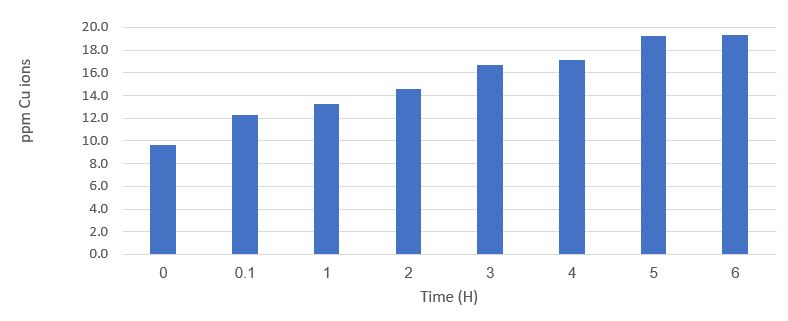
Figure 3 shows the results of time tracking of the concentration of copper ions (Cu2+ + Cu+).
Figure 3: Monitoring the concentration of copper ions over time after applying a voltage of 0.3V between the electrodes.
The results shown in Figure 3 show that the concentration of copper ions almost doubles and remains constant after doubling. These results support the process:
Cu+2 +e à Cu+ E0=+0.159V (cathode reaction)
Cu0 à Cu+ +e E0=-0.52V (anode reaction)
Cu+2 + Cu(0) à 2 Cu+ E0=-0.36V.
The process will stop when there are no more divalent copper ions in the solution.
At this point, all the divalent copper ions have been reduced, and in return, the same amount of monovalent copper ions will be obtained from the oxidation of copper. The concentration of the ions will increase by a factor of 2. Tracking the current gives full compliance with the Faraday constant. At first, the current is relatively high and eventually reaches almost zero.
The above experiments were conducted with a variety of electrodes while maintaining constant experimental parameters. Table 1 shows the times for complete eradication of E. Coli bacteria in the above system.
Table 1
Operating conditions: Initial concentration of copper ions 1ppm, voltage 0.3V, temperature 30 °C, initial E. coli bacterial population 10000 bacteria per milliliter, electrode dimensions 1 x 3 cm, 1 cm spacing.
| Electrode type
(Material, morphology) |
minutes to 90% reduction in bacterial population (E. coli)
|
| Pure copper, smooth | 180
|
| Pure copper, foamed
|
30 |
| Bronze 7% tin in copper, smooth
|
20 |
| Porous Bronze 7% tin in copper, grains (diameter 50 microns) sintered | 5 |
| Pure silver | 30 |
| Titanium, graphite | No reduction in bacterial population |
| None, without dissolved copper ions | No reduction in bacterial population |
| Pure copper without dissolved copper ions | No reduction in bacterial population |
As mentioned in previous work [2], higher temperature and lower pH have a positive effect on the antibacterial activity of monovalent copper ions. Figure 4 shows results tracking the bacterial population over time at different temperatures, and Figure 5 shows the correlation of pH with time to complete bacterial eradication.
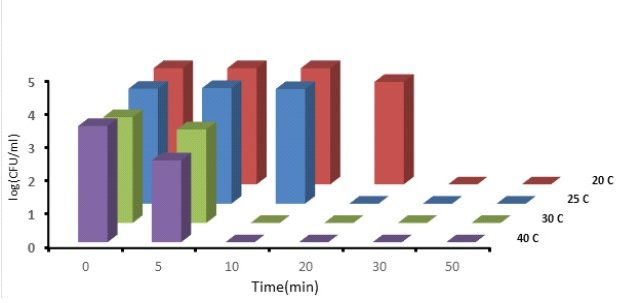
Figure 4: Monitoring the E. coli bacterial population over time, initial concentration of copper ions 0.5ppm, porous bronze electrodes at temperatures of 20, 25, 30, 40 C.
Temperature has a significant effect; the time to kill bacteria is shortened by two times at a temperature of 40 degrees compared to 20 degrees.
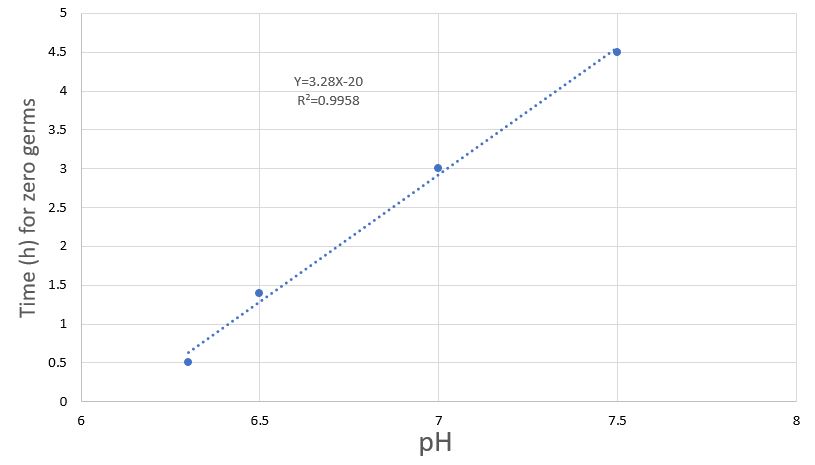
Figure 5: Monitoring time to complete E. coli bacterial eradication, initial concentration of copper ions 0.2ppm, Room temperature, at initial pH of 6.3, 6.5, 7, 7.5.
Consistent with the previous publication, a linear correlation was found between the pH level and the antibacterial activity of monovalent copper.
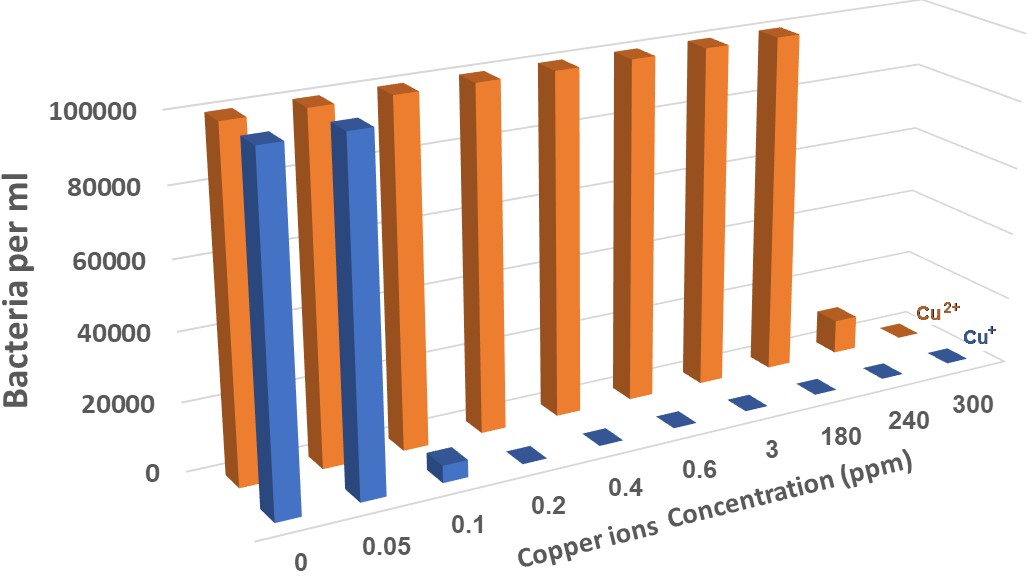
different concentrations of copper ions (Cu+ compared to Cu2+) were tested on 104 E. coli bacteria per milliliter, using Porous Bronze 7% tin in copper, grains (diameter 50 microns) sintered and a constant potential of 0.3V for Cu+ ions, and in the same conditions (temperature of 30 C and pH=6.5) without the electrodes for Cu2+ ions. The resolute present un figure 6.
Figure 6: Monitoring E. coli bacterial population with different copper ion concentrations (Cu+ compared to Cu2+), temperature of 30 °C, and pH=6.5.
It can be concluded from the results shown in Figure 6 that even a concentration of 0.1 ppm of monovalent copper ions has a bactericidal effect, while divalent copper requires a concentration 2400 times greater to obtain the same result.
Apart from the Escherichia coli bacteria, the method was successfully tested on: Staphylococcus aureus, Bacillus thuringiensis, Enterobacter aerogenes, Micrococcus luteus, Staphylococcus epidermidis, Streptococcus faecalis, Pseudomonas aeruginosa, Delftiatsuruhatensis, Staphylococcus cohnii, Brevibacillus brevis, Cyanobacteria, Brewer’s yeast, baking yeast, Macrophomina phaseolina, and Aspergillus prasiliensis.
Pool experiments (up to 20 m3)
To implement the technology in large volumes, a circulation system was developed. First, the water passes through a cation exchanger loaded with divalent copper ions; the cations in the water exchange the divalent copper ions according to the equilibrium constants.
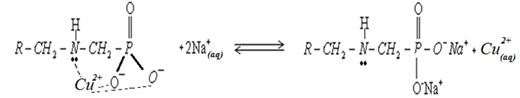
The water with the divalent copper ions (about 0.2ppm) continues into an electrochemical cell built of porous bronze electrodes in a coaxial geometry so that the water flows through the electrodes to obtain a large surface area for electrochemical activity. The voltage between the electrodes is kept constant (0.3V).
Figure 6 shows a schematic description of the system.
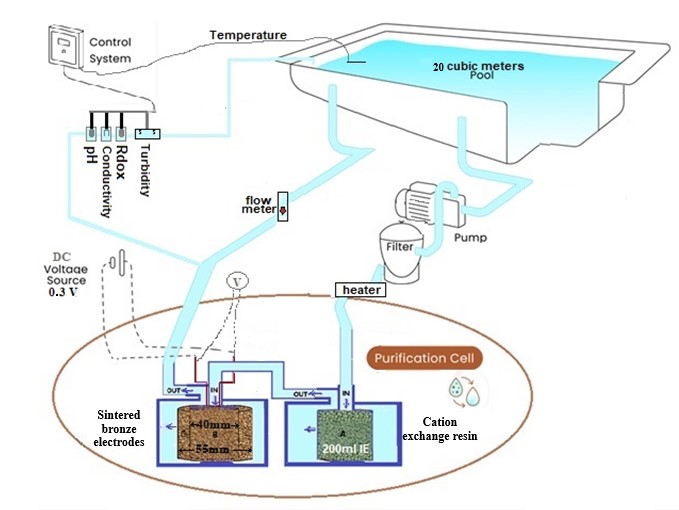
Figure 6: Schematic description of a pool disinfect system
The variation in the CFU/ml bacterial count over time in days is shown in Figure 7. Under normal conditions in the pool, a quantity of zero bacteria per milliliter was measured throughout the duration of the experiment. The experiments involved the deliberate introduction of bacteria into the pool and measuring the time required for their quantity to decrease to zero bacteria.
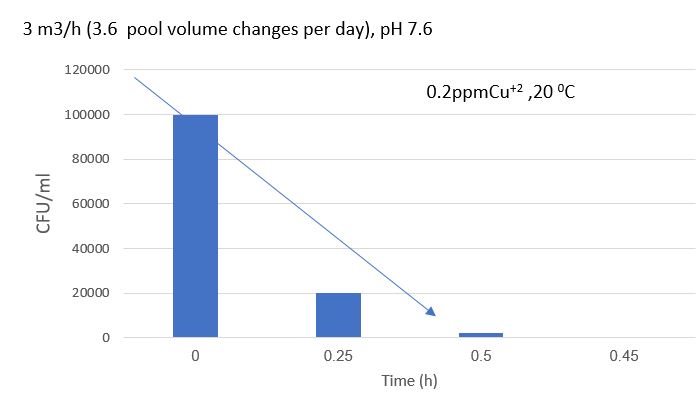
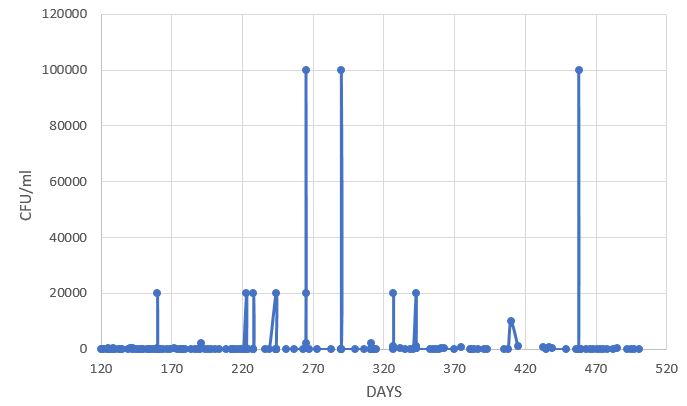
Figure 7: Bacteria (E.Coli) count per milliliter (y-axis- CFU/ml) as a function of the time the pool was operated (Down). you can see the initial introduction of bacteria into the pool and the reaching of zero bacteria in less than 30 min (Up).
The above results are the result of an experiment lasting more than a year (2023-24). During the above experiment, pH, ORP, conductivity, temperature, turbidity, and copper ion concentration were also monitored.
Table 2
| pH | Voltage
Across electrodes (V) |
Turbidity
(NTU) |
Conductivity
(mS) |
Redox potential
(mV vs. Ag/AgCl)
|
|
| Average | 7.52 | 0.3 | 0.7 | 0.68 | 250 |
| Acceptable
Range [18] |
6.5-7.8 | 0.28 – 0.32 | 0.4-1 | 0.5-1.5 | 180-320 |
In addition to the collection of samples on an almost daily basis and the CFU/ml counts reported in Figure 7, the effect of the treatment on other microorganisms was studied. On three occasions during the 12 months, samples were collected and tested to determine levels of certain microorganisms as tabulated in Table 3 (average of the three measurements).
Table 3
| Organism | Average Result | Test method |
| Legionella | <100 CFU/liter | ISO 11731-2:2017 (E) |
| Coliform bacteria | <1 CFU/100mL | SM 9222B |
| Escherichia coli | <1 CFU/100mL | SM 9222G |
| Pseudomonas aeruginosa | <1CFU/100mL | SM 9213E |
| Staphylococcus aureus | <1 CFU/100mL | SM 9213B |
Wastewater disinfection (up to 1m3/h)
We have successfully tested the above technology for wastewater disinfection (sintered sewage after primary treatment to remove solids and secondary treatment to oxidize organic materials using a biotechnological method).
Following laboratory research, a small demonstration facility was established for continuous tertiary treatment of wastewater. A schematic description of the facility appears in Figure 8.
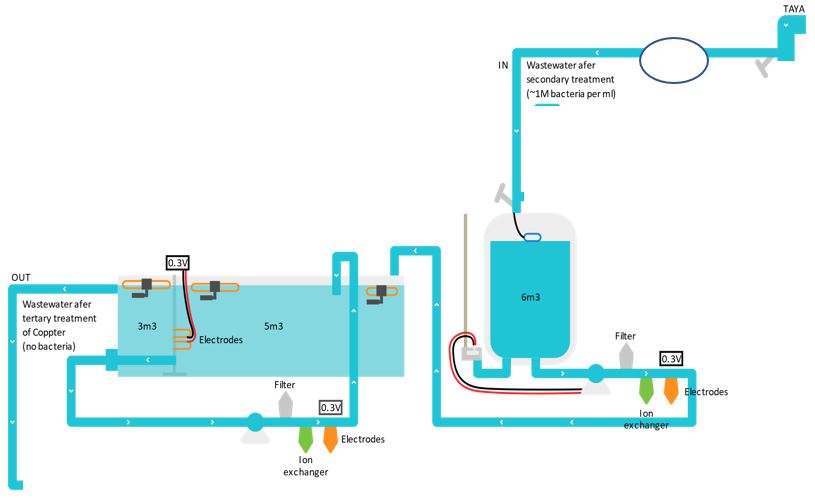
Figure 8: Schematic description of the continuous wastewater disinfection facility with a flow rate of 1 cubic meter per hour.
The process begins with a sand filter that also contains an ion exchanger charged with copper ions to absorb sulfides and the removal of particles over 70 microns. After the filter, the wastewater is pumped into a receiving tank. From the receiving tank, using a pump, the wastewater is pumped at a constant flow rate through a first system of a cation exchanger loaded with copper ions and an electrochemical cell to reduce the divalent copper ions to monovalent, into a circulating intermediate tank. Wastewater is circulated in the tank through an additional system of cation exchanger and an electrochemical cell that ensures effective concentration of monovalent copper ions in the tank, for approximately 5 hours, ensuring complete disinfection of the water. The water from the intermediate tank passes through additional electrodes to the product tank.
The facility operated for approximately 12 months; Table 4 shows the results of the last six months of operation.
Table 4
| MAXIMAL
Bacteria per ml |
MINIMAL
Bacteria per ml |
AVERAGE
Bacteria per ml |
|
| In | 100,000 | 10,000 | 1,000,000 |
| Out | 15 | 0 | 100 |
The concentration of copper ions at the outlet was monitored throughout the operation and did not exceed 0.2ppm.
In the first months of operation, the system suffered from several problems, the most serious of which was waves of sulfides in the wastewater, which, even at low concentrations, precipitate the copper ions.
Conclusions
This study demonstrates several methods for taking advantage of the recognition that copper’s antimicrobial activity is chiefly due to its monovalent copper ions. We have extended our previous research (studied the antimicrobial effects of Cu+ in specialized conditions) to conditions closer to ordinary room environments, using substances and methods that can serve practical applications. We demonstrated the ability to disinfect water without compromising its quality for bathing, agriculture, or drinking.
References