The results presented in this article demonstrate that monovalent copper ions at low concentrations, at which bacteria and fungi are eradicated, do not harm human suspended cells, help tissue healing, and maintain skin vitality.
Abstract
The results presented in this article demonstrate that monovalent copper ions at low concentrations, at which bacteria and fungi are eradicated, do not harm human suspended cells, help tissue healing, and maintain skin vitality.
The monovalent copper ion is unstable in an aqueous environment due to self-oxidation and reduction to divalent and metallic copper, as well as rapid reaction with molecular oxygen. Since it is difficult to create a durable concentration of monovalent copper in physiological solutions, the effect of this ion on cells and tissues has hardly been studied until now.
In recent years, the effect of monovalent copper ions on bacteria and fungi has been investigated [1,2,3]. The results show a dramatic bacteriostatic and fungistatic effect; 0.2 mM of monovalent copper ions can eliminate a population of more than a million bacteria per milliliter at a temperature of 30 °C within minutes.
In addition, studies have found a significant effect of monovalent copper ions on enzymes; monovalent copper ions inhibit (non-competitively) enzymes such as DNA polymerase and phosphatase, thus offering a possible explanation for the antibacterial mechanism [4].
In those studies [1,2,3,4], methods for working with significant concentrations of monovalent copper were developed and applied in this study.
In this study, the effect of monovalent copper ions on vitality, the Scratch test, cell migration, and skin tissue vitality (in vitro) was tested.
It was found that at relatively low concentrations, monovalent copper ions constitute a trigger for a morphological change that causes the cell to detach from the substrate. This detachment allows the cell to migrate to a suitable empty spot (such as a wound). Such migration is part of a wound-healing mechanism. Thus, the findings indicate that monovalent copper ions at concentrations that eradicate microorganisms do not cause damage to human cell vitality. Moreover, at relatively low concentrations, monovalent copper ions even aid in wound closure, as emerged from the test in which cells were scratched and the closure of the distance between cells was tracked over time (Scratch test), and preserves human skin tissue compared to soap, which damages it.
Keywords: Copper (I) ions; Antibacterial effect; Enzyme inhibitor; suspension of human cells; Scratch test; cells vitality; mitochondria; wound closure, skin tissue vitality (in vitro).
Copper (Cu) is an essential trace element for all living organisms [5]. In high concentrations, however, it can exert a biocidal effect that is often lethal for microorganisms [6].
The use of copper compounds to kill microorganisms has been common since ancient times. There is evidence that in ancient Egypt and ancient cultures, and as early as 2200 BC, copper was used to purify drinking water, treat skin diseases, wounds, infections, and more. In the 18th century, copper was widely used clinically to treat lung diseases in Europe [7]. An investigation of copper toxicity on microorganisms began around 1960; it was found that copper surfaces biocides and growth media containing copper reduce the spread of viruses, yeasts, and bacteria [8,9].
Monovalent copper ion is rare in nature due to a disproportionate reaction that forms metallic copper and divalent copper from two monovalent copper ions, and, in addition, molecular oxygen, oxidizes it rapidly [10,11].
Recently, we suggested that monovalent copper ions (Cu(I)) are the active factor in copper’s antimicrobial activity of copper [1]. Under anaerobic conditions and stabilizers (like ascorbic acid and acetonitrile), the effect is stronger than Ag(I), which is also known as an antimicrobial agent [1]. In addition, we found that under the following conditions –of an acidic pH, a carbon source less favored by bacteria, of elevated temperatures and low oxygen levels – the antibacterial action of Cu(I) ions is significantly boosted for wide classes of bacteria and yeasts in vitro [2]. Still, the mechanism of copper toxicity has not been fully understood.
Following studies examining the effect of monovalent copper ions on microorganisms, we will focus on the effect of monovalent copper ions on human lung cells (A549) to further understand their effect and develop future medical solutions.
Anaerobic conditions (see our previous studies [1,2,3,4]):
Production of monovalent copper ions for testing the effect on cell cultures: Monovalent copper ions are almost uncommon in nature due to their sensitivity to oxygen. Therefore, to prepare monovalent copper, we used the salt of divalent copper (CuCl2). The salt was dissolved in a deaerated solution (5% FCS), containing 1% acetonitrile and a piece of metallic copper. The solution was sealed in an airtight bottle (a bottle of penicillin) that prevents the oxidation of the monovalent copper. After about 24 hours, the Cu(II) reacted with zero copper to obtain 2Cu(I) (according to Equation 1) [12,13].
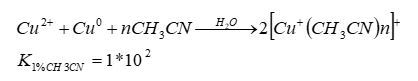
Equation 1
Injection of copper ion solutions via syringes: Solutions containing Cu(I) ions were gently injected through a three-way syringe valve into the medium.
Comparison of the effect of monovalent copper ions: was with ratio to the solution of divalent copper metal ions, divalent nickel, divalent zinc, and the stabilizer (acetonitrile) was dissolved in a 5% FCS medium under aerobic conditions and control (5% FCS).
Cell preparation: A549 human lung epithelial cancer cells were cultured in Institute medium (RPMI-1640) supplemented with 10% FBS, penicillin (100 U/ml), streptomycin (100 μg/ml) and L-Glutamine (2 mM). The cells were maintained in incubator at 37 °C and 5% CO2 in a water-saturated atmosphere. Before the experiment, the cells (7×104 cell/ml in well) were seeded into the 96-well plates and incubated at 37 °C, 5% CO2 overnight. After overnight (16 hours) the plate was taken out of the incubator and checked, and a mono layer of cells was examined under a microscope.
Coloring and counting cells with the help of blue tripene and counting cells:
In this study, trypan blue was used to differentiate between living and dead cells. In dead cells, the color penetrates the cell membrane, which is stained blue, and vice versa, living cells are cells that the dye cannot penetrate through the membrane and are not stained [14]. After 16 hours of cell seeding and examination of a homogeneous layer of cells under a microscope, the growth medium (10% FCS) was removed in each well and a solution with the metal ions was introduced (at a concentration suitable for testing). The plate was placed in the incubator for a predetermined time for living-cells testing. After the test time, the solutions in the wells were removed, and turpentine was added to each of the test wells to detach the cells. After 3 minutes of incubation, a growth medium was added. Using a micropipette, a sample was taken from the well and mixed with an equivalent amount of trypan-blue. 10 µl from the mixture was taken to a counting surface, and the living and dead cells were counted in a solution, using the device EVE ™ Automated Cell Counter.
“Scratch” test: “Scratch” test allows for quantitative and qualitative analysis of cell migration in vitro and analysis of cell-cell interactions and cell with respect to cell migration. Cell migration is an important process whose mechanism of action affects many and varied physiological aspects, such as the wound healing process, in which cell migration plays a key role in epithelialization, a process in which epithelial cells cover the wound and cause it to heal [17,18]. In this study, after 16 hours from the end of sowing the cells on the plate, the plate was removed from the incubator and examined under a microscope to check for a homogeneous layer of cells. A “Scratch” was performed with a tip measuring 200 µl along the well. The growth medium was removed in each well, rinsing was done by adding and pumping a 5% FCS serum preheated for 30 minutes in a water bath at 37°C. At the end of the rinsing, the transition metal ion solutions (in a concentration suitable for testing) were added to each well and a photo of the wells was taken and monitored under a microscope and the “ImageJ” software at predetermined times.
Skin tissue vitality (in vitro): Assessing skin irritation potential in vitro tested using a Reconstructed Human Epidermis (RhE) model. sample exposure time (18 vs 24 h), SDS positive. Resulting tissue viability was assessed by MTT reduction, and IL-1α release was assessed by ELISA. Testing was performed using Reconstructed human Epidermis (RhE)” (ISO, 2016). All incubations were performed in a humidified incubator with standard cell culture conditions of 37 °C and 5% CO2. Cell viability after sample exposure was determined using the MTT viability assay [19].
In addition, histological examinations were performed, and microscopic photographs of the skin tissue section were taken.
Statistical processing of the results: All the experiments presented in this article were repeated 5-9 times, and the results in the “Results” section blow are described as mean ± standard deviation.
The influence of Cu(I) ions on cell vitality
The effect of transition metal ions on the vitality of lung epithelial cells in the range of 0.02-2 mM concentrations was examined by staining cells with trypan blue. Figure 1 shows results which examine the effect of the metal ions at 0.2 mM concentration on the cell vitality after 7,24,48 hours of exposure to substances (an effect showing the percentage of cells living during this time relative to zero time).
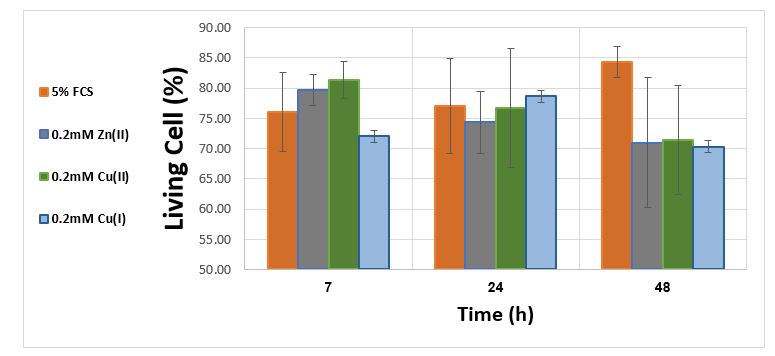
Figure 1. Effect of Cu (I), Cu (II), Ni (II) and Zn (II) at a concentration of 0.2 mM on the count of normal cells per ml normalized to zero time
From the results shown in figure 1 it can be learned that the monovalent copper has no significant toxic effect at concentration of 0.2mM on lungs epithelial cells in vitro.
The influence of Cu(I) ions on detaching cells from the surface
A “trigger” can detach cells from the surface. Figure 2 presents the influence of monovalent copper ions on the phenomenon.
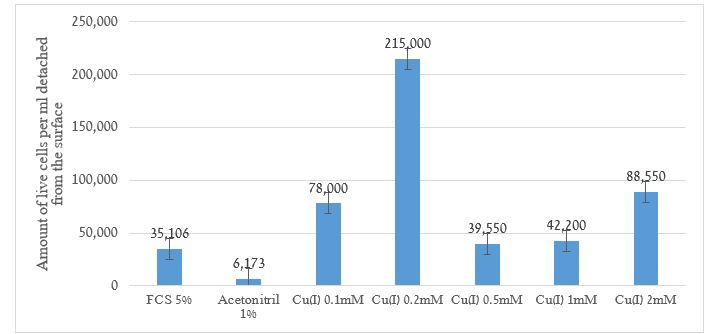
Figure 2. The amount of live cells per ml detached from the surface measured in range of 0.02-2 mM Cu+
The results show a good fit for the “trigger” assumption. The cells detached from the surface. One of the uses of the phenomenon of cell detachment from the surface to which they are attached is cell migration for the purpose of building new tissue in a damaged area.
“Scratch” test in vitro: The scratch assay enables testing cell migration by monitoring tissue gap closure. In Figure 3, a photograph of an experiment in which a scratch creates a gap in lung epithelial cell tissue is shown. The photograph was taken at different times in three sets, the first an audit, the second with the presence of the divalent copper ions, and the third in the presence of a monovalent copper ions.

Figure 3. Photographs of the gap in “Scratch test” experiments in lung epithelial cells tissue in 0,12,24, and 30 hours from the start. The top row shows an audit experiment; the middle row demonstrates an experiment in the presence of 0.2mM Cu2+; and the bottom row shows the effect of gap closing in the presence of 0.2mM Cu+
Figure 4 shows the data for “Scratch” test experiments in the presence of divalent zinc, nickel and copper ions in addition to monovalent copper. The results are normalized in percent with the initial gap set at 100%.
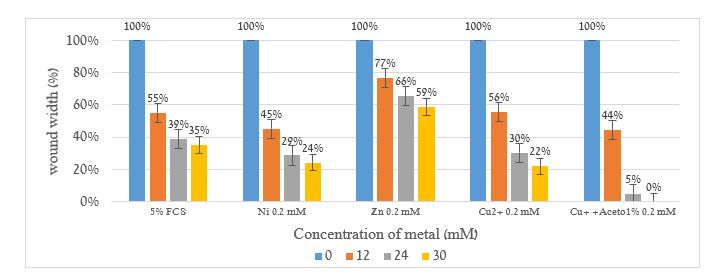
Figure 4. “Scratch” test experiments in lungs epithelial cells tissue in 0,12,24 and 30 hours from the start in the presence of 0.2mM Zn2+, Ni2+, Cu2+ and Cu+ ions. The results are normalized in percent with the initial gap set at 100%.
It is clear from the results shown in figures 3 and 4 that monovalent copper ions cause the cells to close the gap faster than the control and Zn2+, Ni2+, Cu2+ ions.Figures 5 and 6 show skin tissue vitality (in vitro), including a viability test (MTT), skin irritation assessment (IL-1α release), and histological examinations.
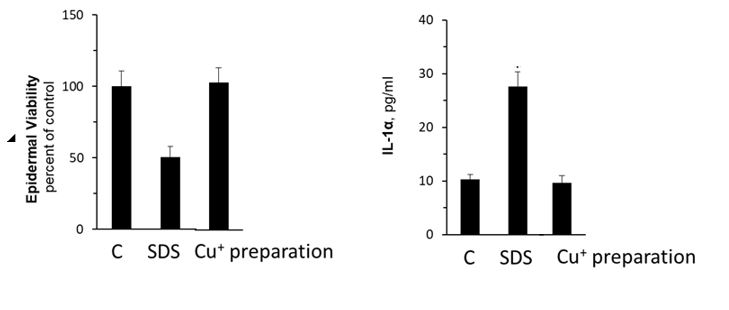
Figure 5. Epidermal Viability using MTT viability assay (Left) and skin irritation assessment measuring IL-1α release (right) for control tissue, SDS (Sodium dodecyl sulfate) as positive control, and Cu+ ion preparation treated tissue.
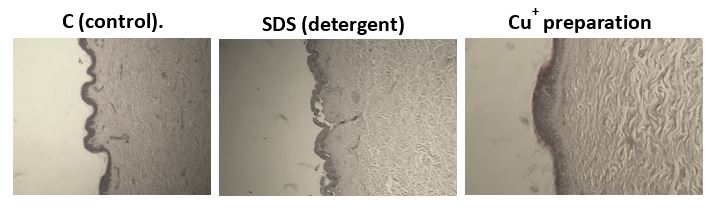
Figure 6. Microscopic photographs of the skin tissue section, left- control, middle- SDS, Right Cu+ ion preparation treated tissue
From the results, it can be concluded that monovalent copper ions do not damage skin cells and do not cause skin irritation; histological examinations show that monovalent copper ions maintain intact and healthy skin tissue.
In this study, we tried to examine the effects of monovalent copper ions have on human lung epithelial cells and skin tissue.
As can be seen from Figure 1, the effect of monovalent copper ions (0.2mM Cu+, Lethal Concentration for bacteria and fungi) on the cell’s vitality is relatively low. Most human cells survive. This result is better than the effect of antibiotics and antifungal preparations used for medical treatment.
The influence of Cu(I) ions on detaching cells from the surface (Figure 2) show a good fit for the “trigger” assumption. Cells detached from the surface. Result of morphological changes of the cells and detachment from the surface were observed in L929 mouse fibroblasts cells [20] and UMR106 cells [21].
One of the uses of the phenomenon of cell detachment from the surface to which they are attached is cell migration for the purpose of building new tissue in a damaged area.
“Scratch” test allows testing cell migration by surveying tissue gap closure. This is a good measure to estimate wound healing factors.
It is clear from the results (Figure 3, 4) that monovalent copper ions cause the cells to close the gap faster than the control.
Tests on human skin tissue (in vitro) show that monovalent copper ions do not damage epithelial cells or induce sensitization (Figure 5). The skin tissue maintains its integrity, as can be seen in a macroscopic cross-section photograph of the skin samples (Figure 6).
These results are in line with other studies which found that copper ions contribute to wound healing [22, 23]. These studies have attributed the healing ability of copper to its influence on proteins that encourage intensive metabolism, angiogenesis, cell migration, over-epithelialization and more, as shown in Figure 8 [22].
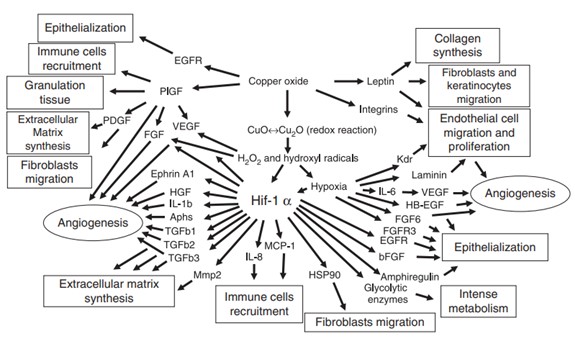
Figure 8. Proposed mechanisms by which copper oxide stimulates angiogenesis and wound healing [22]
The findings indicate that monovalent copper ions at concentrations that eradicate microorganisms do not cause damage to human cell vitality. Moreover, at relatively low concentrations (Lethal Concentration for bacteria and fungi), monovalent copper ions even aid in wound closure.